NUCALA FOR CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) WITH AN EOSINOPHILIC PHENOTYPE
FOR ADULT PATIENTS

Proven efficacy and established safety across 5 indications
NUCALA FOR CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) WITH AN EOSINOPHILIC PHENOTYPE
FOR ADULT PATIENTS

NUCALA FOR SEVERE EOSINOPHILIC ASTHMA (SEA)
FOR AGES 6+

NUCALA FOR CHRONIC RHINOSINUSITIS WITH NASAL POLYPS (CRSwNP)
FOR ADULT PATIENTS

NUCALA FOR EOSINOPHILIC GRANULOMATOSIS WITH POLYANGIITIS (EGPA)
FOR ADULT PATIENTS

NUCALA FOR HYPEREOSINOPHILIC SYNDROME (HES)
FOR AGES 12+

NUCALA is for the:
An anti-IL-5 approved for select eosinophil-driven diseases | 8:58
In this NUCALA video series, hear from specialists in immunology, pulmonology, critical care medicine, and otolaryngology.
Watch more expert videos:
NUCALA in patients with SEAkeyboard_arrow_right
Navigate a NUCALA prescription
Review NUCALA patient profiles and dosing information

TYPES OF PATIENTS WHO MAY BENEFIT FROM NUCALA
These profiles may remind you of patients in your practice.
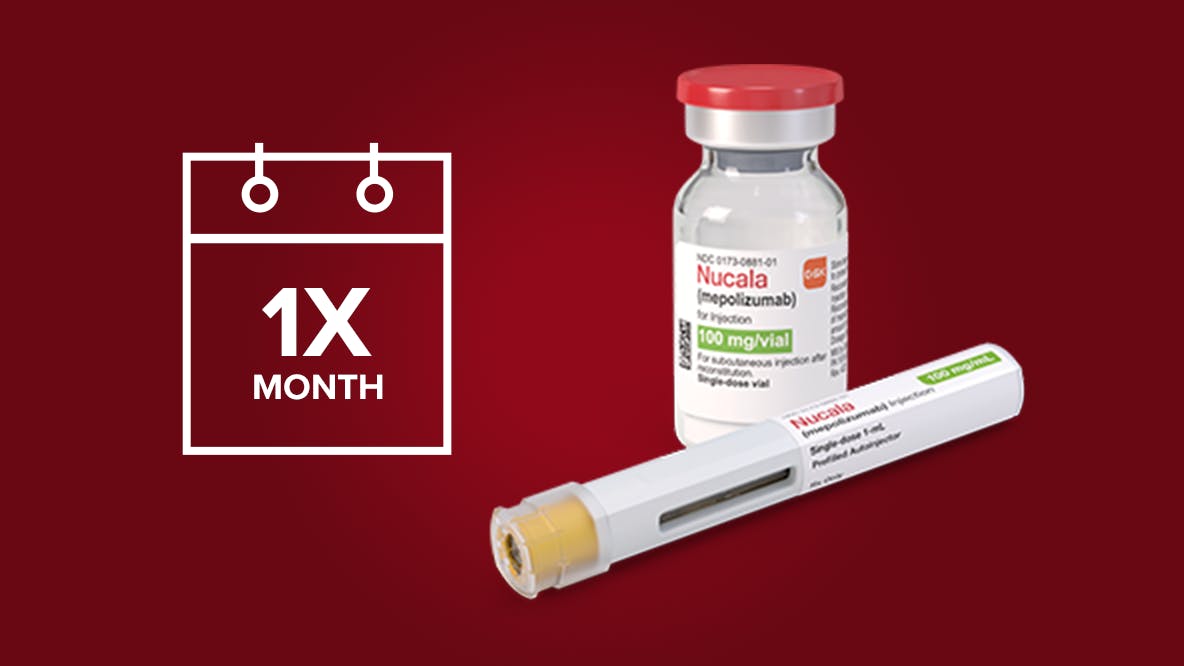
FLEXIBLE ADMINISTRATION OPTIONS FOR ONCE-MONTHLY* NUCALA DOSING
Offer patients fixed dosing independent of weight, and the option of at-home or in-office administration.
*Every 4 weeks.
Explore data and patient access details
BLOOD EOSINOPHIL REDUCTION WITH NUCALA
NUCALA reduced blood eosinophils and maintained them within normal levels across indications.1,2 The mechanism of action of mepolizumab has not been definitively established.
Note: Mean normal blood eosinophil levels in a general population of subjects without confounding factors have been reported as 100 cells/µL (range: 34-257) and 107 cells/µL (5th, 95th percentile: 30, 395); levels vary based on age, sex, environmental exposures, and comorbid conditions.1,2
NUCALA HAS BROAD NATIONAL COVERAGE, WITH 95% OF COMMERCIAL PATIENTS COVERED NATIONALLY†
Individual access may vary by geography and plan benefit design.
Formulary status may vary and is subject to change. Formulary coverage does not imply clinical efficacy or safety. This is not a guarantee of partial or full coverage or payment. Consumers may be responsible for varying out-of-pocket costs based on an individual’s plan and its benefit design. Each plan administrator determines actual benefits and out-of-pocket costs per its plan policies. Verify coverage with plan sponsor.
†Pharmacy benefit coverage as of March 2026. “Patients” means covered lives for all approved indications across commercial and employer payer types (excluding Managed Medicaid) as calculated by Managed Markets Insight & Technology (MMIT), LLC.
Source: MMIT, LLC database as of March 2026.
Covered=Any potential for reimbursement from a health plan and may include step edits, prior authorizations, and other restrictions.